We leverage systems biology to make fundamental contributions to human health. We do both basic and applied science, with a major focus on translating our science to positively impact society. We are interested in the optimization of wellness, the minimization of disease, the reversal of disease in its earliest stages, the precise diagnosis of disease when it occurs, and the development of treatments. These are all deeply data-driven pursuits, and we have spent the past several years developing unique systems to approach each of these challenges. We see medicine as being at a fundamental tipping point now, transforming from a reactive disease-care system to a proactive discipline of health maintenance as well. We believe systems medicine is poised now to make huge advances. Below are a few of the major areas of effort we are engaged in. We hope that many of you will join in these endeavors.
1. The 100,000 Person Wellness Project (100K Wellness Project)
A major focus in the lab currently is the 100K Wellness Project. This project was initiated jointly by Dr. Lee Hood and Dr. Nathan Price (see e.g. Hood and Price, Science Translational Medicine, 2014), and represents a pioneering study to create a data-rich, longitudinal study of sufficient magnitude and depth to identify the early transition states for the common human diseases. The project also focuses on the development of quantifiable metrics of wellness to better understand how to optimize wellness for the individual. Indeed, we see the focus on wellness as being the key to understanding the earliest deviations into disease, where it can hopefully be avoided or reversed. Certainly, many cases show that this is possible, and that much suffering from disease can be avoided if caught and dealt with at an early stage before irreversible damage is done. We launched this effort in 2014 with the Hundred Person Wellness Project (or Pioneer 100), which provided concrete proof-of-concept for this approach. The results of this study will be submitted for publication shortly (Price, N.D. et al., In preparation).
This focus on wellness has led us to launch a new sector: Scientific Wellness. The central step in this effort was the creation of Arivale, which was co-founded by Dr. Lee Hood, Dr. Nathan Price, and Clayton Lewis (who is serving as CEO). ISB has a partnership with Arivale where — for all participants who consent to let their data be used anonymously for research purposes — we can do deep dive analyses based on these data in order to invent the future of scientific wellness and proactive P4 Medicine (P4 = Predictive, Preventive, Personalized, and Participatory). These data include whole genome sequencing on everyone, and then (multiple times per year) measurements out of the blood (clinical chemistries, metabolomics, selected proteins), stool (microbiome), and saliva (cortisol stress tests), as well as continuous measurements based on wearable digital health devices (e.g. FitBit).
Dr. Price is leading the analytical efforts for the 100K Wellness Project, and this partnership provides us with data of unprecedented scale and depth to create the future of Scientific Wellness and an approach to Medicine that is Predictive, Preventive, Personalized, and Participatory (P4).
2. Systems Medicine
We develop and use systems approaches to disease. This involves analysis and generation of high-throughput omics data to search for diagnostics and therapeutic targets. We have a strong focus on neurodegenerative diseases, cancers, and, recently, pre-term birth. The uniforming principles of our systems medicine efforts is the analysis of emergent phenomenon and the leveraging of large amounts of interconnected data streams to search for patterns and mechanisms that are highly differentiating of the disease and (where data available) on its initiation and progression.
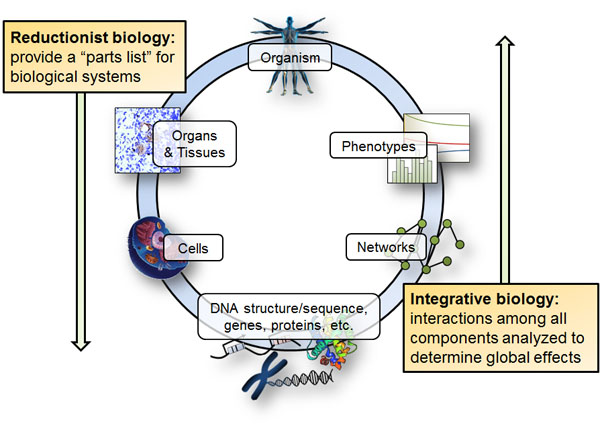
3. Network analysis
Towards the goal of understanding, diagnosing, treating, and preventing human disease (and sometimes towards environmental concerns), the Price lab is using an approach grounded in biomolecular network analysis. We use a continuum of approaches ranging from the fully mechanistic to the purely statistical as needed—no one method works best for all cases. One large divide exists between network models that are rooted in biochemistry and those that are driven by statistics, where the underlying biochemistry is often unknown. Integrating these network models into a unified computable model is essential for predictive interrogation of the genotype-to-phenotype relationship. Our work on networks is mechanistically deepest for metabolic and transcriptional regulatory networks, though (especially with statistical approaches) we can leverage essentially any biological network type. Bridging statistical and mechanistic network models provides a powerful means to deeply integrate disparate types and multiple scales of data. Such approaches are timely because they combine abundant omics data sets (e.g., genomics, transcriptomics, metabolomics and proteomics) with established biochemical/mechanistic networks in order to study the effects of genomic (or other) perturbations in disease.


 hood-price.isbscience.org/research/research-overview/
hood-price.isbscience.org/research/research-overview/
